Molecules to metabolism
* FALSIFICATION OF THEORIES: THE ARTIFICIAL SYNTHESIS OF UREA HELPED TO FALSIFY VITALISM.
- Vitalism states that organic compounds in plants and animals can only be synthesized with the help of a vital principle that exists in the organism itself. The origin and phenomena of life can't be physically or chemically forced.
- In 1928, Wöhler synthesized urea artificially by heating ammonium cyanate.
* UREA AS AN EXAMPLE THAT IS PRODUCED BY LIVING ORGANISMS BUT CAN ALSO BE ARTIFICIALLY SYNTHESIZED.
- Urea is synthesized naturally when there is an excess of amino acids in the body.
- Chemical reactions catalysed by enzymes happen to make urea.
- Urea is transported to the kidney and is passed out of the body in urine.
- Can be synthesized artificially by heating a solution of silver cyanate and ammonium chloride to produce ammonium cyanate (and silver chloride).
- Heating the ammonium cyanate causes its atoms to rearrange, ansforming into urea.
* MOLECULAR BIOLOGY EXPLAINS LIVING PROCESSES IN TERMS OF THE CHEMICAL SUBSTANCES INVOLVED.
- Studies the interactions between molecular components that carry out different biological processes in living cells.
- Nucleic acids consists of DNA and RNA. Thesse chemicals are used to make genes and then proteins.
- Molecular components make up biochemical pathways that provide cells with energy, processes information from outside the cell, generate new DNA and replicate the cellular DNA genome.
* CARBON ATOMS CAN FORM FOUR BONDS ALLOWING A DIVERSITY OF COMPOUNDS TO EXIST.
- Carbon atoms can form up to four, strong covalent bonds, making these complex structures very stable.
- Bonds between carbon atoms can be used to make rings or chains.
* LIFE IS BASED ON CARBON COMPOUNDS INCLUDING CARBOHYDRATES, LIPIDS, PROTEINS AND NUCLEIC ACIDS.
- Living organisms use four main classes of carbon compunds with different properties to serve different purposes.
- Carbohydrates are composed of the elements C, H and O. There are two hydrogen atoms for every oxygen atom. Includes glucose, fructose, starch, cellulose and sucrose.
- Lipids are a broad class of C, H and O elements which is insouble in water. Triglycerides are fats if solid at room temperature and oils if liquid at room temperature. Includes wax, fatty acids, steroidsand triglycerides.
- Proteins are composed of one or more chains of amino acids, containing C, H, O and N elements. Two of the twenty amino acids contains sulfur. Includes haemoglobin, insulin, amylase, pepsin, antibodies and adrenaline.
- Nucleic acids are composed of chains of nucleotides, containing C, H, O, N and P elements. The two types of nucleic acids are ribonucleic acid (RNA) and deoxyribonucleic acid (DNA).
* DRAWING MOLECULAR DIAGRAMS OF GLUCOSE, RIBOSE, A SATURATED FATTY ACID AND A GENERALIZED AMINO ACID.
- Hydroxyl: —OH
- Amine: —NH₂
- Carboxyl: —COOH
- Methyl: —CH3
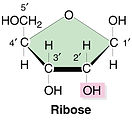
- Ribose (a simple sugar) has the formula C₅H₁₀O₅
- The molecule is a five-membered ring with a side chain.
- Four carbon atoms are in the ring and one forms the side chain.
- The carbon atoms can be numbered starting with number 1 on the right.
- The hydroxyl groups (OH) on carbon atoms 1, 2 and 3 point up, down and down respectively.
- Glucose has the formula C6H12O6.
- The molecule is a six-membered ring with a side chain.
- Five carbon atoms are in the ring and one forms the side chain.
- The carbon atoms can be numbered starting with number 1 on the right.
- The hydroxyl groups on carbon atoms 1, 2, 3 and 4 point down, down, up and down respectively.
- Saturated fatty acids consists of an unbranched chain of carbon atoms.
- There are usually 14 - 20 carbon atoms.
- At one end of the chain the carbon atom is part of carboxyl group.
- At another end, the carbon atom is bonded to three hydrogen atoms.
- All other carbon atoms are bonded to two hydrogen atoms.
- They only form single covalent bonds.
- Monounsaturated fatty acids have one double bond.
- Polyunsaturated fatty acids have more than one double bond.
- Amino acids consists of a carbon atom in the centre of the molecule. It makes four bonds with other things:
- An amine group, hence the term amino acid.
- A carboxyl group which makes the molecule an acid.
- A hydrogen atom.
- The R group, which is the variable part of amino acids.
* IDENTIFICATION OF BIOCHEMICALS SUCH AS CARBOHYDRATE, LIPID OR PROTEIN FROM MOLECULAR DIAGRAMS.
- Proteins contain nitrogen and sometimes sulfur where as carbohydrats and lipids don't.
- In carbohydrates, there are two hydrogen atoms for every oxygen atom.
- Lipids contain relatively less oxygen atoms than carbohydrates.
* METABOLISM IS THE WEB OF ALL THE ENZYME CATALYZED REACTIONS IN A CELL OR ORGANISM.
- The sum of all chemical reactions in an organism.
- Consists of pathways where a molecule is transformed intoanother.
* ANABOLISM IS THE SYNTHESIS OF COMPLEX MOLECULES FROM SIMPLER MOLECULE INCLUDING THE FORMATION OF MACROMOLECULES FROM MONOMERS BY CONDENSATION REACTIONS.
- Anabolism requires energy, usually in the form of ATP.
- Protein synthesis including ribosomes.
- DNA synthesis during replication.
- Photosynthesis, including production of glucose from carbon dioxide and water.
- Synthesis of complex carbohydrates including starch, cellulose and glycogen.
* CATABOLISM IS THE BREAKDOWN OF COMPLEX MOLECULES INTO SIMPLER MOLECULES INCLUDING HYDROLYSIS OF MACROMOLECULES INTO MONOMERS.
- Catabolism releases energy and is captured in the form of ATP, which can then be used in the cell.
- Digestion of food in the mouth, stomach and intestines.
- Cell respiration in which glucose of lipids are oxidized to carbon dioxide and water.
- Digestion of complex carbon compounds in dead organic matter by decomposers.