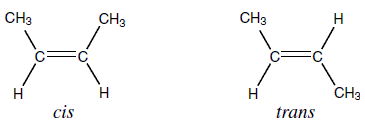Carbohydrates and lipids
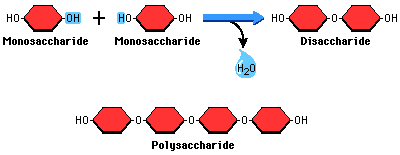
* MONOSACCHARIDE MONOMERS ARE LINKED TOGETHER BY CONDENSATION REACTIONS TO FORM DISACCHARIDES AND POLYSACCHARIDE POLYMERS.
- Glucose, fructose and ribose are examples of monosaccharides. They can be linked together to make larger molecules.
- Monosaccharides are single sugar units.
- Disaccharides consist of 2 monosaccharides linked together. (Maltose is made by linking 2 glucose molecules, sucrose is made by linking a fructose and glucose).
- Polysaccharides consist of many linked monosaccharides. These include starch, cellulose and glycogen- all made of glucose molecules.
- Monosaccharides combine through condensation where an OH is lost from one molecule and H is lost from the other. It yields water.
- Condensation polymerization is anabolic so energy in the form of ATP is supplied to the monosaccharides to use.
- The remaining oxygen atom after water has been removed is the glycosidc linkage. It is between the 1st and 4th carbon atom.
- Hydrolysis is the opposite of condensation polymerization where polysaccharides break down upon reaction with water.
* STRUCTURE AND FUNCTION OF CELLULOSE AND STARCH IN PLANTS AND GLYCOGEN IN HUMANS.
- Starch, glucogen and cellulose are all composed of glucose but the type of glucose and linkage results in their differences.
Glucose
- Has 5 OH groups, any can be used in condensation reactions but only 3 of them are used to link to make polysaccarides.
- Most common link is between the OH on carbon atom 1 and 4.
- The OH on carbon atom 6 is used to form side branches.
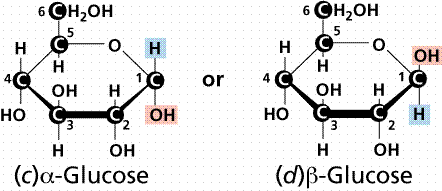
- In alpha glucose, the OH group points downwards whereas in beta glucose, the OH group points upwards.
Cellulose
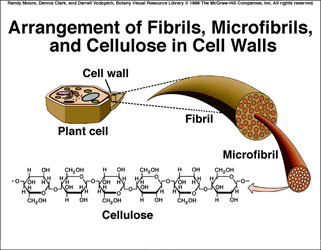
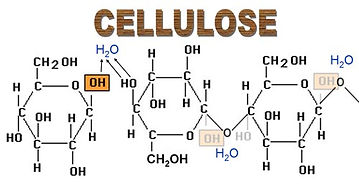
- Made by linking together Beta glucose molecules.
- Condensation reactions link carbon atom 1 to carbon atom 4 of the next Beta glucose. They are orientated alternately upwards and downwards so the OH and H molecules can combine to yield water.
- Cellulose molecule is therefore a straight chain.
- They are unbranched chains of Beta glucose. Can form bundles (microfibrils) with hydrogen bonds which links the cellulose molecules together.
Starch
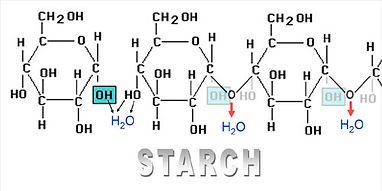
- Made by linking together Alpha glucose molecules.
- Condensation reactions link carbon atom 1 to carbon atom 4 of the next Beta glucose.
- The OH groups point downwards (orientated in the same way) so the starch molecule is curved.
- Two forms of starch: Amylose is a chain of unbranched Alpha glucose molecules and forms a helix. Amylopectin is branched and has a more globular chape.
- Only made by plant cells. Starch is hydrophillic but too large to dissolve in water. Useful for storing large amounts of glucose (as high glucose concentrations result in water entering the cells).
- Used as a store of energy in seeds and storage organs such as potato organs. Also a temporary store in leaf cells when more glucose is made by photosynthesis than it can be exported to other parts of the plant.
Glycogen
- Glycogen is a more branched form of starch so the molecule is more compact.
- Made by animals and some fungi.
- Stored in the liver and some muscles in humans.
- Acts as a store of energy in the form of glucose (prevents osmotic problems).
- Easy to add or remove extra glucose molecules to both starch and glycogen. Can be done at either ends of a branched or unbranched molecule.
- Starch and glycogen do not have a fixed size (number of glucose molecules can change).
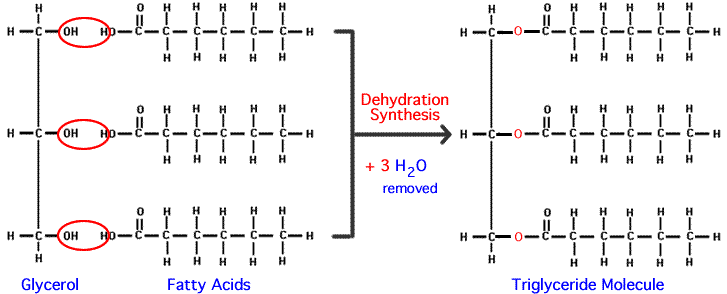
* TRIGLYCERIDES ARE FORMED BY CINDENSATION FROM THREE FATTY ACIDS AND ONE GLYCEROL.
- Triglycerides are one of the principal groups of lipid. Oil in sunflower seeds and fat in adipose tissue in humans are examples.
- Fats are liquid at body temperature and solid at room temperature. Oils are liquid at both temperatures.
- An ester bond is formed where a fatty acid is linked to the glycerol.
- Ester bonds are formed when an acid reacts wuth the OH group on an alcohol. Here, the reaction is between the COOH group on a fatty acid and an OH on the glycerol.
- Used as energy stores which can be released by aerobic cell respiration.
- They don't conduct well and are used as heat insulators (blubber of Arctic marine mammals).
* LIPIDS ARE MORE SUITABLE FOR LONG TERM ENERGY STORAGE IN HUMANS THAN CARBOHYDRATES.
Lipids
- Lipids that are used are fats. They are stored in the adipose tissue, located immediately beneath the skin and around some organs like the kidneys.
- The amount of energy released in cell respiration per gram of lipids is double the amount of carbohydrates.
- The same amount of energy stored as lipid rather than carbohydrates therefore adds half as much to the body mass. Fats form pure droplets in cells with no water associated whereas each gram of glycogen is associated with 2 grams of water so lipids are actually six times more efficient in the amount of energy that can be stored per gram of body mass.
- Mass of energy storage medium is important as we have to carry them around. Even more important in animals which can fly.
- Stored lipids have secondary roles that can't be performed as well by carbohydrates. Lipids can be used as heat insulators (also why stored fat is right under skin).
- As it is liquid at body temperature, fats can act as a shock absorber (also why it is found around vital organs).
Carbohydrates
- Glycogen (carbohydrate) is used for energy storage in the liver and some muscles.
- It can be broken down to glucose rapidly, then transported easily by the blood to the are which needs it. Fats in the adipose tissue can't be mobilized as rapidly.
- Glucose can be used anaerobically or aerobically whereas fats and fatty acids can only be used in aerobic respiration.
- The liver stores up to 150 grams of glycogen. Muscles store up to 2% glycogen by mass.
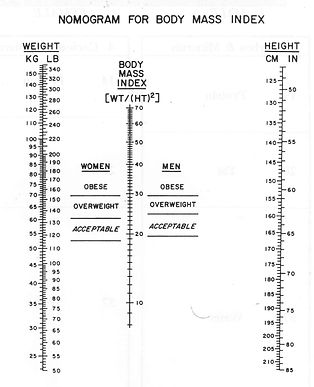
* DETERMINATION OF BODY MASS INDEX BY CALCULATION OR USE OF A NOMOGRAM.
- BMI was developed by Adolphe Quetelet (Belgian statician).
- BMI = mass in kilograms
height in meters squaered
- Units for BMI are kg/m-2
- A nomogram is a chart shows 3 straight lines- height and body mass on either sides of the BMI line.
- Where food supplies are insufficient or unevenly distributed, people may become underweight.
- Anorexia nervosa is a psycological condition that involve volutary starvation and loss of body mass.
- Where food supplies calorie intake are in excess, people may become obese.
- Skinfold callipers can be used to estimate the amount of body fat.
- Obesity increases the risk of coronary heart disease and type 2 diabetes.
- Obesity reduces life expectancy significantly and is increasing the overall costs of healthcare.
* FATTY ACIDS CAN BE SATURATED, MONOSATURATED OR POLYSATURATED.
- Structure: A chain of carbon atoms with hydrogen atoms linked to them by single covalent bonds. There is an acidic, carboxyl group (COOH) at one end.
- Most fatty acids have between 14 and 20 carbon atoms. Length is variable.
- Carbon atoms may be linked by single covalent bonds. Each carbon atom can bond with two hydrogen atoms (as much as they possibly can), hence the name, saturated fatty acid.
- Carbon atoms may be linked by double covalent bonds. Each carbon atom can only bond with one hydrogen atom ( contains less hydrogen than they could), and these are unsaturated.
- Monosaturated fatty acid contains one double bond whilst a polyunsaturated fatty acid contains multiple double bonds.
* UNSATURATED FATTY ACIDS CAN BE CIS OR TRANS ISOMERS.
- If the hydroen atoms are nearly always on the same side of the two carbon atoms that are double bonded, they are called cis-fatty acids.
- If the hydrogen atoms are on alternating sides of the carbon atoms, they are called trans-fatty acids.
- The chain of cis-fatty acids is bent at the double bond. This makes it harder for these to be packed in regular arrays, and results in a lower melting point. Triglycerides with cis-unsaturated fatty acids are therefore usually liquid at toom temperature.
- Trans-fatty acids do not bend at the double bond and is solid at room temperature. These are produced artificially produced by partial hydrogenation of vegetable or fish oils (solid fats for use in margarine and other processed foods).
* SCIENTIFIC EVIDENCE FOR HEALTH RISKS OF TRANS-FATS AND SATURATED FATS.
- Coronary heart disease is caused when coronary arteries become partially blocked by fatty deposits, leading to blood clot and heart attacks.
- There is a positive correlation between fatty acid intake and rate of CHD, but no actual evidence to prove saturated fats causes CHD. It could be due to low amouns of dietary fibre that causes it.
- In Maasai (Kenya), people's diet consists of high levels of fatty acid (milk, meat) intake but CHD is rare.
- Olive oil contains cis-monosaturated fatty acids and is eaten in many countries around the Mediterranean but has low rates of CHD. It could be due to genetics or other factors like the common use of tomatoes in dishes that lower the rate of it.
- There is a positive correlation between intake of trans-fat and rates of CHD. Other factors did not appear to account for the correlation so trans-fats probably causes CHD. Patients who have died from CHD have high concentrations of trans-fats in their diseased arteries.
* EVALUATING CLAIMS: HEALTH CLAIMS MADE ABOUT LIPIDS NEED TO BE ASSESSED.
- Health claims about food (healthy or not) can be tested scientifically on laboratory animals. with the same, age, sex and state of health. Variables such as temperature, amount of exercise and diet can be controlled so they do not influence the results of the experiment. If only one dietary factor varies, strong evidence can be obtained about the effects of this factor on the animal.
- Effects on human may be different but it is hard to perform experiments on them. Unless they are identical twins, the humans would be genetically different (even if they are the same age and sex). It is hard to control variable such as exercise and diet of an individual for an extended period of time.
- Evidence is therefore obtained by epidemiological studies. A large roup of people will be studied- measuring their food intake and following their health over a period of years. Statistical procedures are used to see whether factors in the diet are associated with a higher frequency of a particular disease (factors that could cause the disease need to be eliminated).
* NATURE OF SCIENCE QUESTION: USING VOLUNTEERS IN EXPERIMENTS.
- During WW2, volunteers were willing to sacrifice their health to help extend medical knowledge in the US and England.
- A vitamin C experiment was conducted where 20 volunteers were given a daily dose of 70mg of vitamin for six weeks. Then for the next eight months, three volunteers stayed at 70mg a day, seven on 10mg a day and ten on 0mg a day. Those recieving no vitamin C developed scurvy. Their wounds would not heal and they were bleeding from the hair follicles & gums. Some even suffered from severe heart problems.
- The same experiment was conducted on guinea pigs who cannot sythesize ascorbic acid. They were killed afterwards and the ones recieving less or no vitamin C had less cross-linking between the protein fibres in the collagen of the bone, therefore having less strength.
- Is it ethically acceptable for doctors or scientists to perform experiments on volunteers, where there is a risk that the health of the volunteers will be harmed?
- Sometimes people are paid to participate in medical experiments, such as drug trials. Is this more or less acceptable than using unpaid volunteers?
- Is it better to use animals for experiments or are the ethical objections the same as with humans?
- Is it acceptable to kill animals, so that an experiment can be done?
* EVALUATION OF EVIDENCE AND THE METHODS USED TO OBTAIN THE EVIDENCE FOR HEALTH CLAIMS MADE ABOUT LIPIDS.
- An evaluation is the assessment of implications and limitations. Evidence for health claims come from scientific research.
- Implications: Do the results of the research support the health claim strongly, moderately or not at all?
- Limitations: Were the research methods used rigorous or are there uncertainties in the conclusion because of weaknesses in methodology?
Implications: We can analyze the results (easier if results are presented visually or as a graph)
- Is there a correlation (positve or negative) between the intake of lipid being investigated and the rate of the disease / health benefit?
- How large is the difference between the average rate of disease with different levels of intake? Small differences may not be significant.
- How widely spread is the data? The more widely spread the data is, the less significant the average rate it.
- If statistical have been done on the data, do they show significant differences?
Limitations: We can assess the methods used.
- How large is the sample size? Surveys usually need to be carried out on thousands of people in order to get reliable results.
- How even was the sample in age, sex, state of health and lifestyle? The more even, the less other factors can affect the results.
- If the samling was uneven, were there adjustments made in the experiments to eliminate the effects of the other factors?
- Were the measurements of lipid intake and disease rate reliable? Volunteers may not report their intake accurately diseases are sometimes are misdiagonized.